Checkmate 8162/8/2024 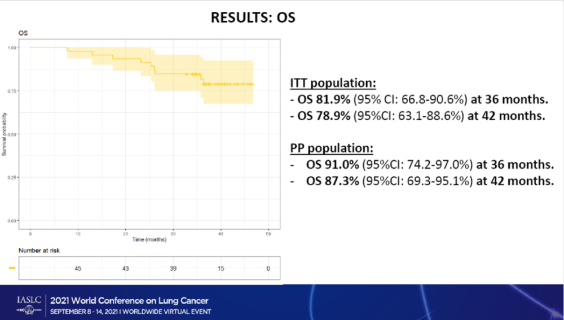
Opdivo -based treatments have now shown benefit in four Phase 3 clinical trials in early-stage cancers, including lung cancer, bladder cancer, esophageal/gastroesophageal junction cancer and melanomaīristol Myers Squibb (NYSE: BMY) today announced that the Phase 3 CheckMate -816 trial met a primary endpoint of pathologic complete response (pCR) in resectable non-small cell lung cancer (NSCLC). Positive results mark the first time an immune checkpoint inhibitor-based combination has demonstrated superior efficacy versus chemotherapy as neoadjuvant therapy in a Phase 3 trial in resectable non-small cell lung cancer 
Clinical Trial Registration: NCT02998528 ().Ĭlinical trial immunotherapy lay summary neoadjuvant treatment nivolumab non–small cell lung cancer plain language summary presurgery treatment.CheckMate - 816 met a primary endpoint of improved pathologic complete response in patients who received Opdivo plus chemotherapy before surgery Results from CheckMate 816 support the benefit of using nivolumab plus chemotherapy before surgery for people with resectable NSCLC. Adding nivolumab to chemotherapy did not lead to an increase in the rate of side effects compared with chemotherapy alone, and side effects were generally mild and manageable. Most people went on to have surgery in both treatment groups the people who took nivolumab plus chemotherapy instead of chemotherapy alone had less extensive surgeries and were more likely to have good outcomes after less extensive surgeries. More people in the nivolumab plus chemotherapy group had no cancer cells left in the tumor and lymph nodes removed by surgery. Researchers found that people who took nivolumab plus chemotherapy lived longer without the cancer getting worse or coming back compared with those who took chemotherapy alone. The researchers also assessed how adding nivolumab to chemotherapy affected the timing and outcomes of surgery and whether the combination of these drugs was safe. The researchers assessed whether people who received nivolumab plus chemotherapy lived longer without the cancer geting worse or coming back and whether there were any cancer cells left in the tumor and lymph nodes removed by surgery. In total, 179 people were randomly assigned to receive nivolumab plus chemotherapy, and 179 people were randomly assigned to receive chemotherapy alone. During this study, a computer randomly assigned the treatment each person would receive before surgery for NSCLC. The goal of CheckMate 816 was to find out if nivolumab, an immunotherapy that activates a person's immune system (the body's natural defense system) to fight cancer, plus chemotherapy works better than chemotherapy alone when given before surgery in people with non-small-cell lung cancer (NSCLC) that can be removed surgically (resectable NSCLC).Īdults who had not previously taken medications to treat NSCLC and whose cancer could be removed with surgery were included in CheckMate 816. In this article, we summarize results from the ongoing phase 3 CheckMate 816 clinical study that were published in The New England Journal of Medicine in 2022. 19 Institut du Thorax Curie-Montsouris, Institut Curie, Paris, France. 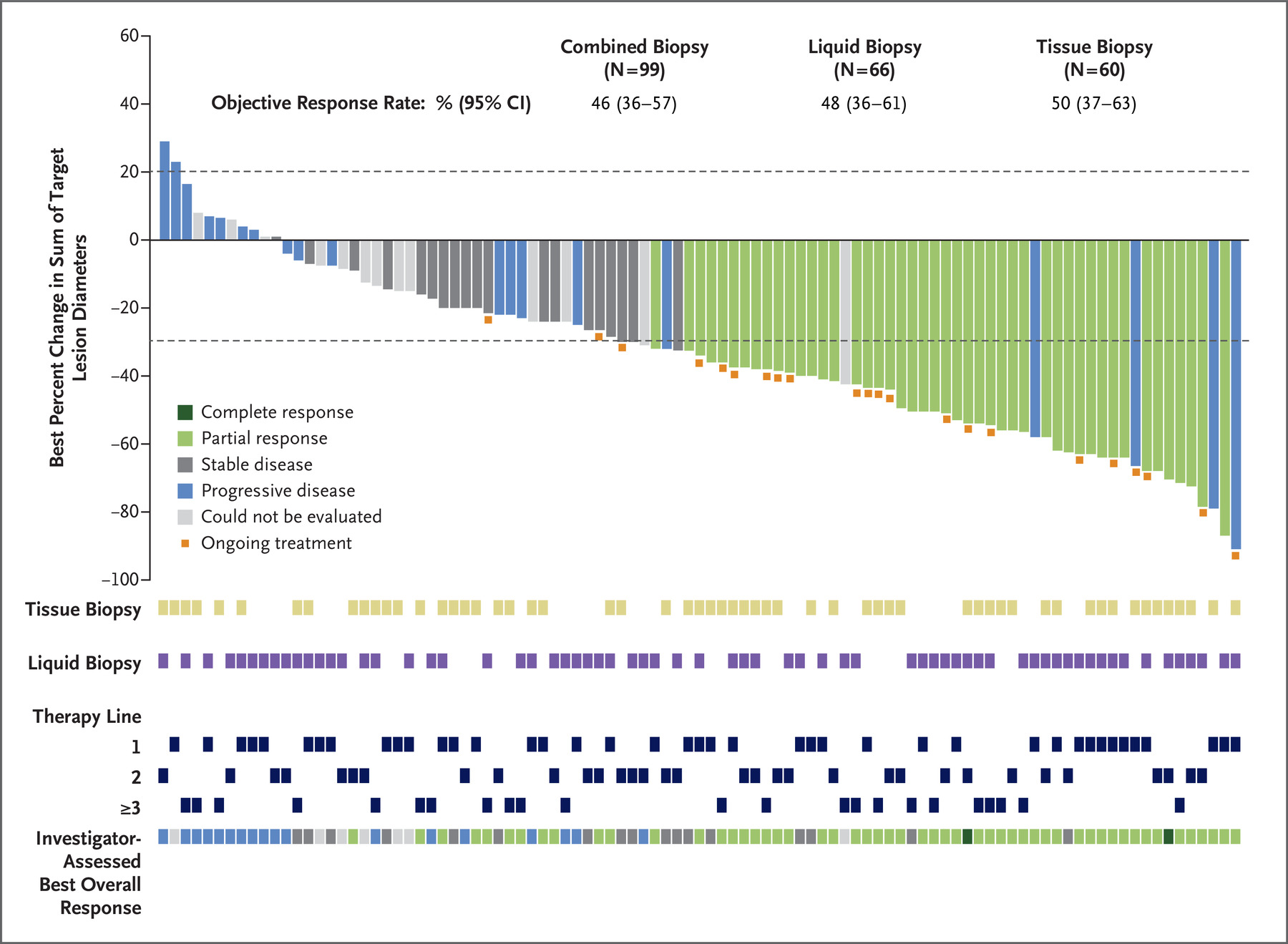
18 Bristol Myers Squibb, Princeton, NJ, USA.17 Bristol Myers Squibb, Braine-L'Alleud, Belgium.16 University of Chicago Medicine, Chicago, IL, USA.15 Centre Hospitalier de l'Université de Montréal, Montréal, Québec, Canada.14 Peking University School of Oncology, Beijing Cancer Hospital, Beijing, China.13 Kanagawa Cancer Center, Yokohama, Japan.12 University of Occupational & Environmental Health, Kitakyushu, Japan.11 Charleston Oncology, Charleston, SC, USA.Ion Chiricuta & Universitatea de Medicina si Farmacie Iuliu Hatieganu, Cluj-Napoca, Romania. 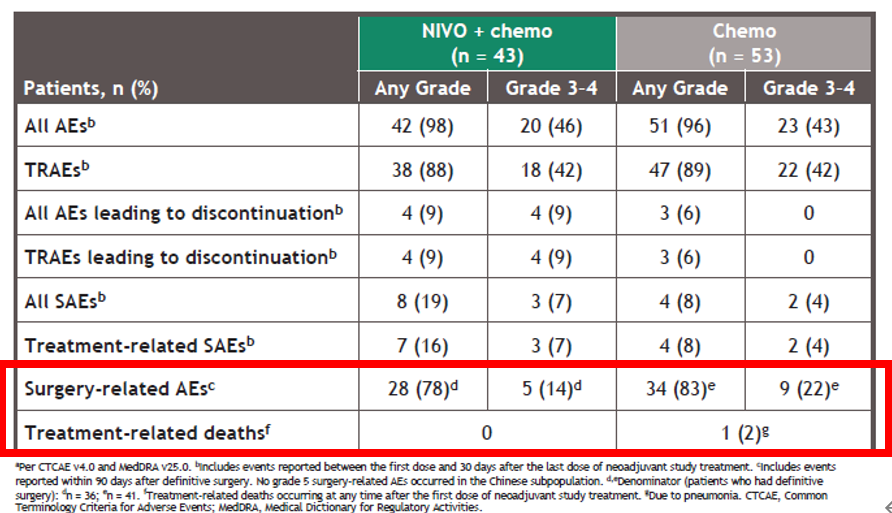
9 Tianjin Lung Cancer Center, Tianjin Medical University Cancer Institute & Hospital, Tianjin, China.8 Aberdeen Royal Infirmary, Aberdeen, United Kingdom.7 Vall d'Hebron Institute of Oncology, Barcelona, Spain.6 Dana-Farber Cancer Institute, Boston, MA, USA.5 Kindai University Faculty of Medicine, Ohno-Higashi, Osaka-Sayama, Osaka, Japan.4 Hospital Universitario Puerta de Hierro, Madrid, Spain.3 Shanghai Chest Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China.2 McGill University Health Center, Montréal, Québec, Canada.1 Bloomberg-Kimmel Institute for Cancer Immunotherapy, Johns Hopkins Kimmel Cancer Center, Baltimore, MD, USA.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
